[ad_1]
Researchers in the United States conducted a study showing that the 2019 coronavirus disease (COVID-19) vaccines developed by Pfizer-BioNTech and Moderna generate a coordinated adaptive immune response capable of eliciting booster responses to future infection with severe acute respiratory syndrome coronavirus. 2 (SARS-CoV-2).
The University of California team found that immunization with Pfizer-BioNTech’s BNT162b2 vaccine or Moderna’s mRNA-1273 induces spike-specific B cell enrichment.
The viral spike protein mediates the initial stage of the SARS-CoV-2 infection process and is the primary target for antibodies after natural infection or vaccination.
Ilhem Messaoudi and colleagues claim that the vaccination also activated CD4 T cells and induces robust antigen-specific polyfunctional CD4 T cell responses.
In addition, cloned expanded CD8 T cells were observed in all vaccine recipients.
A pre-printed version of the research paper is available on the website bioRxiv* server, while the article is subject to peer review.

Vaccination and infection provide two different pathways to immunity
Since the start of the COVID-19 epidemic in December 2019 in Wuhan, China, intense global efforts to rapidly develop effective vaccines against the causative agent of SARS-CoV-2 have led to authorization for use. emergency multiple vaccines.
These include the messenger RNA (mRNA) vaccines developed by Pfizer (BNT162b2) and Moderna (mRNA-1273). The results of clinical trials have indicated that a maximum protection of 95% is achieved within 1 to 2 months after the second dose of either vaccine, including protection against several circulating variants of SARS-CoV- 2 worrying.
However, the mechanisms by which these vaccines elicit long-lasting cellular immune responses to the virus remain poorly understood.
Natural infection and vaccination provide two different pathways to immunity, which studies previously have shown are characterized by distinct T and B cell responses.
“Further studies that integrate functional, transcriptional and repertoire analysis of the response of memory immune cells to COVID-19 mRNA vaccination are needed,” write Messaoudi and colleagues.
What did the researchers do?
The team used single-cell RNA sequencing and functional tests to assess humoral (antibody) and cellular responses to two doses of the Pfizer-BioNTech or Moderna mRNA vaccine (14 days after a second dose) in four people. The results were compared with the immune responses seen in three recovering people who had suffered an asymptomatic or mild infection.
What did the study find?
Infection and vaccination induced anti-SARS-CoV-2 binding and neutralizing antibodies.
These antibodies were detected as early as two weeks after a first dose of vaccine, with levels multiplied by several after a second dose.
While the titers of neutralizing antibodies after the first dose of vaccine were comparable to those of recovering individuals, significantly higher levels were observed in subjects vaccinated after the booster dose.
However, several important adaptations of B lymphocytes have been shared between vaccinees and convalescents.
Flow cytometric analysis revealed a reduction in the level of naive B cells, but an expansion of memory B cells in both groups.
In addition, single-cell RNA sequencing has shown a reduction in IgA + memory B cells after vaccination, a finding that has recently been described for recovering people.
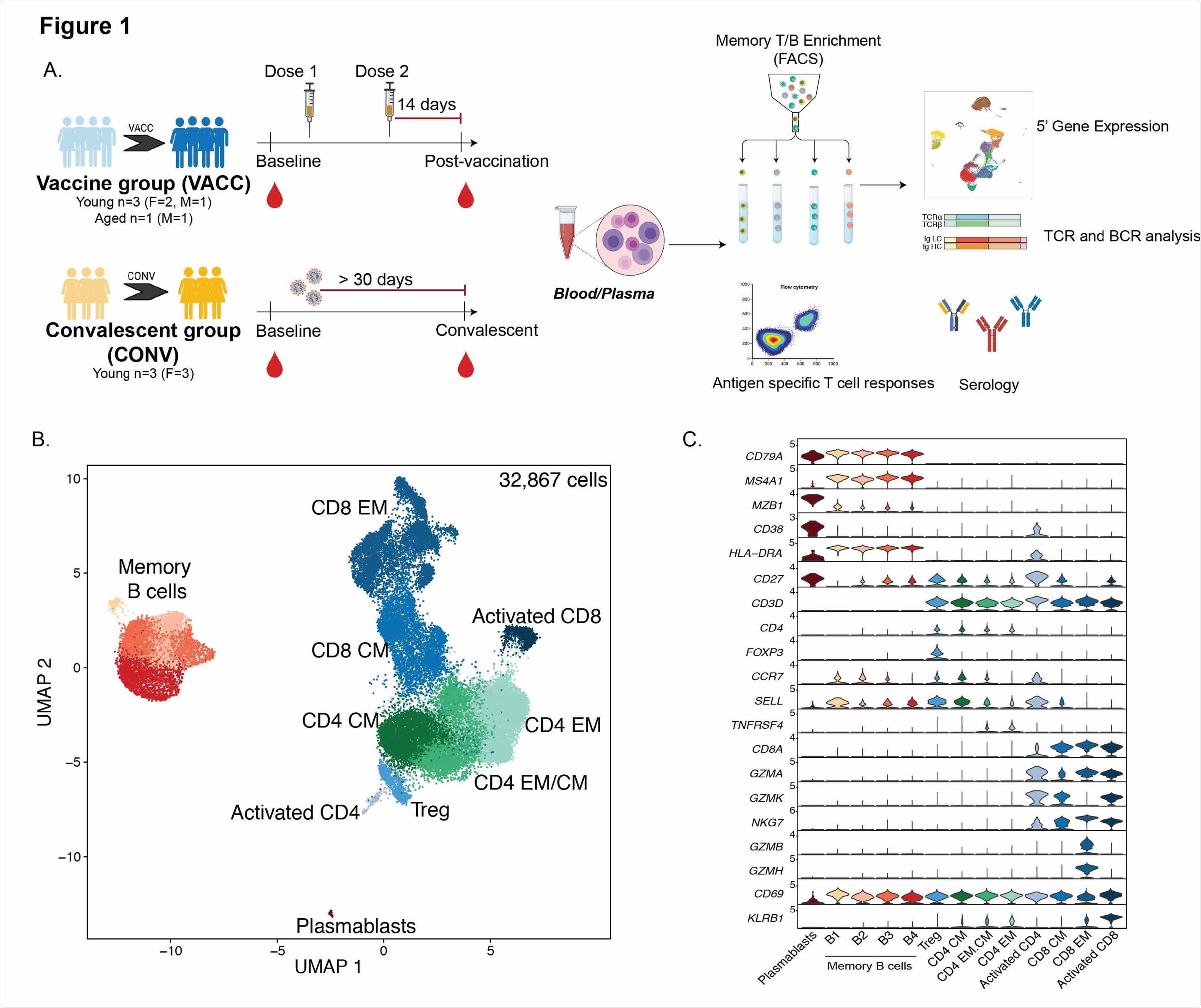
Immunologic changes with SARS-CoV-2 mRNA vaccination (A) Experimental study design. Blood was collected from SARS-CoV-2 naive subjects before vaccination, 2 weeks after dose 1 and 2 weeks after booster vaccination (VACC group) or from individuals exposed to SARS-CoV-2 but asymptomatic (CONV group) before and after convalescence. PBMC immune phenotypes and antigen-specific T and B cell responses were measured using multicolor flow cytometry. Longitudinal serological responses to the vaccine were measured by ELISA and neutralization tests. T and B memory cells from a subset of PBMC samples (n = 4 / group for vaccinated volunteers, n = 3 / group for convalescent, matched health workers) were profiled using baseline scRNA-Seq (pre-vaccination or and after – vaccination time points. (B) UMAP projection of 32,867 T and B memory cells with major subsets annotated. (C) Violin plots of key genetic markers used for cluster annotations Normalized transcription counts are shown on the Y axis.
Vaccination appeared to establish lasting memory and potential recall responses
Importantly, the team observed a modest expansion of plasmablasts and a significant increase in spike-specific B cells only two weeks after a second dose of vaccine, indicating the establishment of lasting memory and booster responses. potential for infection.
Single-cell analyzes also revealed an expansion of activated CD4 + T cells and robust spike-specific polyfunctional CD4 T cell responses after vaccination.
The frequencies of activated CD8 T cells were comparable between the vaccinated and convalescent groups. However, natural infection induced the expansion of larger CD8 T cell clones, including distinct clusters.
This is likely due to the recognition of a larger set of epitopes presented by the virus that is not found in mRNA vaccines, the researchers say.
“Our study highlights a coordinated adaptive immune response where early CD4 T lymphocyte responses facilitate the development of the B lymphocyte response and substantial expansion of effector CD8 T lymphocytes, together capable of contributing to future booster responses,” concludes the team.
*Important Notice
bioRxiv publishes preliminary scientific reports that are not peer reviewed and, therefore, should not be considered conclusive, guide clinical practice / health-related behavior, or treated as established information.
[ad_2]
Source link