[ad_1]
Instead of burning coal and releasing CO2, new research plans to absorb CO2 and produce coal.
A new breakthrough could allow us to burn our coal and have it too. Researchers from Australia, Germany, China and the United States have collaborated on the development of a carbon storage method capable of transforming CO2 gas into solid carbon particles with high efficiency. . Their approach could help us clean up the atmosphere of (some of) the greenhouse emissions we produce – with a hint of style.
Coal idea
"Although we can not literally go back in time, converting carbon dioxide back into coal and burying it in the ground is a bit like raising the pace of emissions," said Torben Daeneke, a member of the Australian Research Council. DECRA and co-author.
The idea of permanently removing CO2 from the atmosphere is not new. In fact, it is widely regarded as a solution to our climate problems caused by ourselves. We have developed a number of methods, but they are simply not yet viable. Current carbon capture technologies convert the gas into a liquid form, which is then transported to be injected underground. However, the process requires high temperatures (which means high costs) and potential leaks from the storage sites cause environmental concerns.
The team's approach, however, is based on an electrochemical technique that captures atmospheric CO2 and transforms it into a solid carbon that is easy to store.
<! – Username: zmescience_300x250_InContent
->
"To date, CO2 has only been converted to a solid at extremely high temperatures, making it unviable on an industrial scale," says Daeneke. "By using liquid metals as a catalyst, we have shown that it is possible to reconvert the gas to carbon at room temperature, according to an efficient and scalable process."
"While it is necessary to continue research, it is a crucial first step in ensuring solid carbon storage."
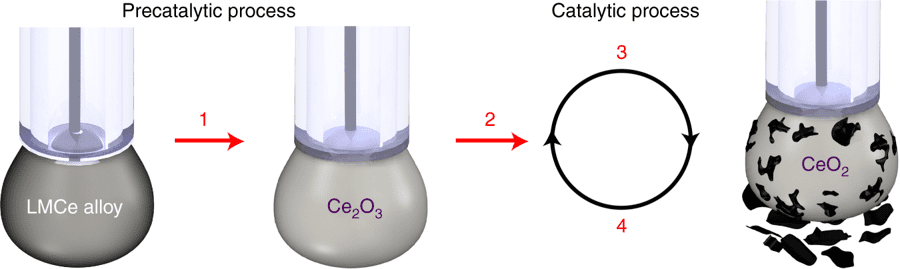
The cerium and liquid metal catalyst has certain surface properties which make it a very good electrical conductor – the current also chemically activates the surface of the catalyst.

Schematic of the catalytic process.
Image credits Dorna Esrafilzadeh, (2019), Nature.
The entire process begins with the dissolution of the carbon dioxide in a beaker filled with liquid and a small amount of liquid metal. When charged with electric current, this catalyst slowly starts to convert the CO2 into solid carbon flakes on its surface and falls off quickly so that the process can be maintained indefinitely.
"One of the benefits of the process is that carbon can hold an electrical charge and become a supercapacitor. It could be used as a component in future vehicles, "says Dr. Dorna Esrafilzadeh, a research fellow at RMIT's Faculty of Engineering. and the main author of the document.
"The process also produces a synthetic fuel as a by-product, which could also have industrial applications."
The document "Reduction of ambient temperature in solid carbon species on liquid metals with thin atomic interfaces in cerium" was published in the journal Nature.
Appreciated this article? Join more than 40,000 subscribers to the ZME Science newsletter. Subscribe now!

[ad_2]
Source link
