[ad_1]
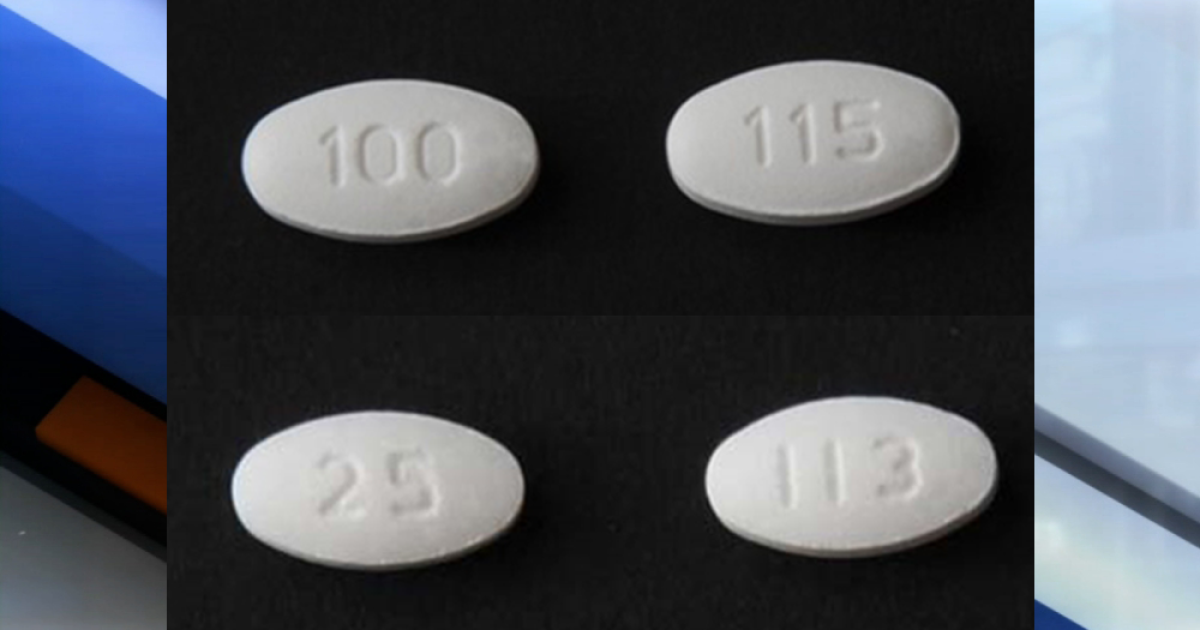
Torrent Pharmaceuticals Limited is extending its recall for losartan potassium tablets and Losartan potassium / hydrochlorothiazide tablets as they contain "N-methylnitrosobutyric acid (NMBA), a potentially carcinogenic acceptable daily intake levels published by the FDA "according to the
Website of the US Food and Drug Administration.
The recall was expanded to include 36 batches of Losartan potassium tablets and 68 Losartan potassium / hydrochlorothiazide tablets. The tablets treat hypertension and hypertensive patients with left ventricular hypertrophy and nephropathy in patients with type 2 diabetes.
For a complete list of tablets affected by the recall,
click here.
Patients "should continue to take their medications because the risk of harm to the patient's health may be higher if the treatment is stopped immediately without further treatment."
Consumers who have medical questions regarding this recall or to report an adverse event can contact Torrent Pharmaceuticals Limited at the following address:
[ad_2]
Source link