[ad_1]
 Copyright of the image
Copyright of the image
Reuters
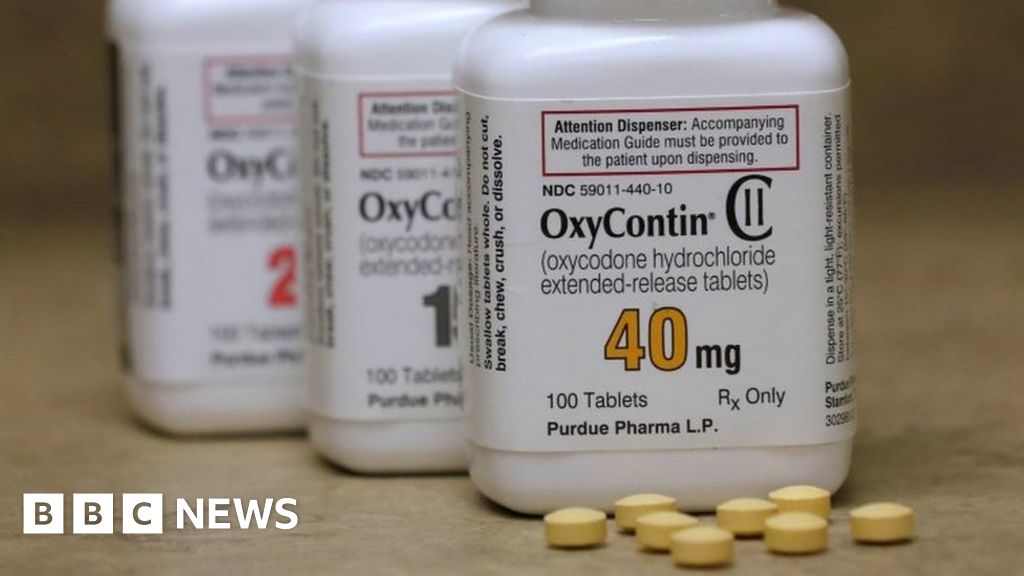
The company has made billions of dollars with products such as OxyContin pain reliever
Drug manufacturing giant Purdue Pharma has reportedly reached a multi-billion dollar interim deal in the US to settle many lawsuits against it.
The company owned by the billionaire family Sackler is accused of helping fuel the opioid crisis in the United States with drugs such as the painkiller OxyContin.
The agreement would remove Purdue from the first federal opioid crisis trial, scheduled to open in Ohio in October.
But society could still face legal battles with states that are not in agreement.
Connecticut, Iowa, Massachusetts, Nevada, New Jersey, New York State, Pennsylvania, North Carolina and Wisconsin are among the states that claim to not be party to Agreement, reports the Associated Press.
The stated regulation should be the largest ever paid by an opioid manufacturer.
What is in the transaction?
Purdue Pharma has negotiated to settle a lawsuit filed by more than 2,000 plaintiffs – including half of the states, local governments and Native American tribes.
The US media reports that the deal would imply that the Sacklers leave the company before it declares bankruptcy, dissolves and reforms.
The profits of the company would then be directed towards the payment to the plaintiffs of an amount – estimated at about 10 to 12 billion dollars – as well as the donation of drugs for the treatment of the dependence and the overdose, according to several reports.
The Sackler family is expected to directly contribute at least $ 3 billion of its personal wealth to the transaction, according to reported information.
According to the Washington Post newspaper, the agreement in principle would have the support of 23 states and about 2,000 local governments – but does not correspond to the national agreement sought by Purdue, according to the Washington Post newspaper.
Copyright of the image
Getty Images
Opioid-related deaths have increased dramatically in recent years
A number of attorneys general have publicly pledged to continue their legal fight against the cabinet.
"This seeming arrangement is a slap for everyone who has had to bury a loved one because of the destruction and greed of this family," said Josh Shapiro, Pennsylvania's senior attorney.
"This allows the Sackler family to ward off the billionaires and admit no wrongdoing."
What is the opioid crisis?
Opioids are a group of drugs ranging from codeine to illicit drugs such as heroin. Prescription opioids are primarily used to relieve pain, but can be highly addictive.
On average, 130 Americans die each day from an opioid overdose, according to the US Center for Disease Control and Prevention, which says more than 200,000 Americans have died from opioid-related overdoses during last two decades.
Purdue is one of the opioid manufacturers, distributors, and pharmacies cited in more than 2,000 lawsuits represented in the federal trial scheduled to begin in Ohio next month.
The cases claim that corporations are responsible for the American crisis of opioid addiction.
Companies such as Purdue are accused of using deceptive practices to sell opioids, including minimizing their addictive quality.

Multimedia playback is not supported on your device
Purdue said the US regulator, the Food and Drug Administration, had approved the labels for OxyContin containing warnings about the risks.
In another lawsuit, a US judge ordered Drugmaker Johnson & Johnson to pay $ 572 million for his contribution to the opioid addiction crisis in Oklahoma last month. Purdue had already settled with the state $ 270 million earlier this year.
Who are the Sackler family?
The brothers Arthur, Mortimer and Raymond Sackler were all doctors in Brooklyn, New York, who in the early 1950s bought a drug company called Purdue Frederick, which would become Purdue Pharma.
Today, the fortune of the Sacklers is estimated at about $ 13 billion. The family is a prolific philanthropist and its name adorns a wing of cultural buildings around the world, including the Louvre in Paris.
While the opioid scandal has engulfed the family, a number of prestigious museums – including the Tate in the UK – have announced that they will no longer withdraw money from the family.
The Sacklers claimed that they were passive members of Purdue Pharma 's board of directors, which had approved routine management requests and had not participated in the marketing of OxyContin.
[ad_2]
Source link