[ad_1]
LIVERPOOL, England, October 31, 2018 – A team from the University of Liverpool is using optical spectroscopy to better understand electrocatalysis, a phenomenon that could lead to more efficient waste conversion, such as carbon dioxide (CO2), usable energy. The team, which develops catalytic systems for sustainable fuel production, uses vibrational frequency generation spectroscopy (VSFG) to study the electrochemical reduction of CO2 in situ and provide insight into the potential of electrocatalysts to convert CO2 in energy-rich byproducts such as carbon monoxide.
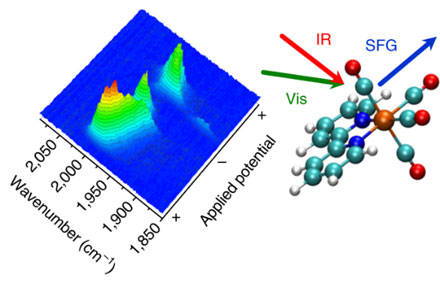
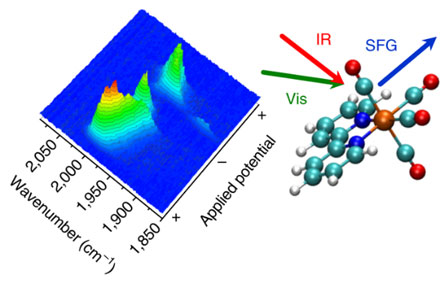
The use of vibrational frequency generation spectroscopy (VSFG) could help scientists to fully exploit the potential of new clean energy technologies. With the kind permission of the University of Liverpool.
Researchers use VSFG, coupled with electrochemical experiments, to explore the chemistry of one of the most promising CO2 reduction electrocatalysts, Mn (bpy) (CO) 3Br. With the help of VSFG, the team was able to observe key intermediates present only at the surface of an electrode for a short time. This had not been possible in previous experimental studies.
Researcher Gaia Neri said, "We have shown that VSFG can track the behavior of even very short-lived species in the catalytic cycle. This is exciting because it provides researchers with new opportunities to better understand the functioning of electrocatalysts, which is an important next step towards commercialization of the electrochemical CO process.2 conversion to clean fuel technologies. "
The team is working to further improve the sensitivity of its technique and is developing a new detection system that will provide a better signal-to-noise ratio.
The search was published in Catalysis of nature (Https://doi.org/10.1038/s41929-018-0169-3).
Source link