[ad_1]
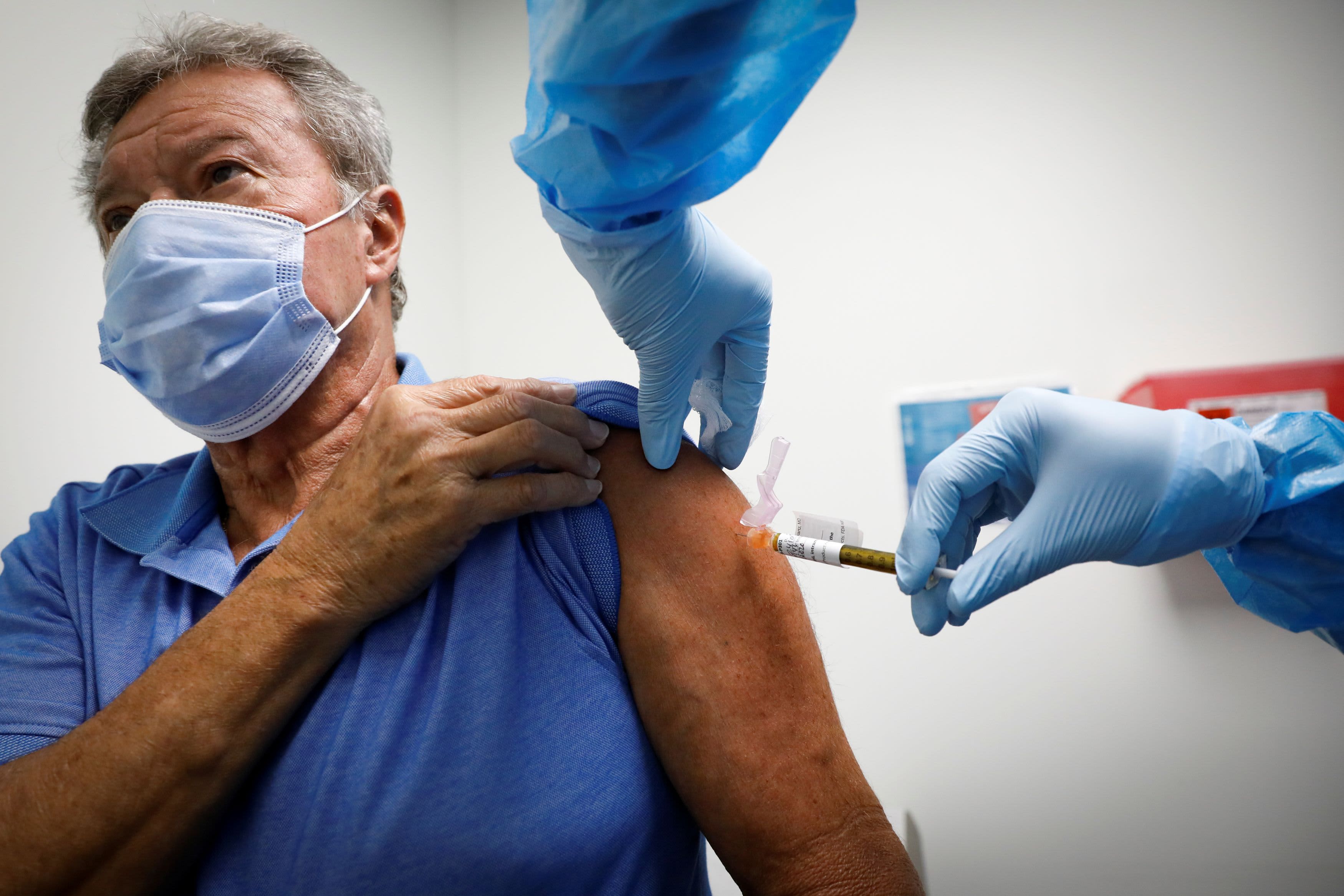
A volunteer receives an injection of a vaccine as he participates in a coronavirus disease (COVID-19) vaccination study at the Research Centers of America, Hollywood, Florida on September 24, 2020.
Marco Bello | Reuters
Public health officials and drugmakers must be transparent about the side effects people may experience after receiving their first coronavirus vaccine, doctors urged during a meeting with CDC advisers on Monday as states are preparing to distribute doses as early as next month.
Dr. Sandra Fryhofer of the American Medical Association noted that Pfizer vaccines and Moderna against Covid-19 require two doses at varying intervals. As a practicing physician, she said she was concerned whether his patients come back for a second dose because of potentially unpleasant side effects they may experience after the first injection.
“We really need to make patients aware that this won’t be a walk in the park,” Fryhofer said in a virtual meeting with the Advisory Committee on Immunization Practices, an external group of medical experts who advise the CDC. She also liaises with the committee. “They are going to know that they got a vaccine. They are probably not going to feel well. But they have to come back for that second dose.”
Participants in Moderna and Pfizer’s coronavirus vaccine trials told CNBC in September they experienced a high fever, body aches, severe headache, day-long exhaustion and other symptoms after receiving injections. While the symptoms were uncomfortable and sometimes intense, participants said they often left after a day, sometimes earlier, and it was better than taking Covid-19.
The two companies acknowledged that their vaccines can induce side effects similar to the symptoms associated with Covid-19 light, such as muscle aches, chills and headaches.
A woman from North Carolina in the Moderna study is in his fifties said she had no fever, but she had suffered a severe migraine that had left her exhausted for a day and unable to concentrate. She said she woke up the next day feeling better after taking Excedrin, but added that Moderna might need to tell people to take a day off after a second dose.
“If this proves to be effective, people are going to have to toughen up,” she said. “The first dose is no problem. And then the second dose will definitely lower you for the day … You will need to take a day off after the second dose.”
During Monday’s meeting, Patsy Stinchfield, a nurse practitioner for children in Minnesota, said officials and drug makers may try to talk some side effects more positively. She said they could use language such as “response” instead of “adverse reaction”.
“These are immune responses,” said Stinchfield, a former voting member of the committee. “And so, if you feel anything after the vaccination, you should expect to feel that way. When you do, it is normal to have arm pain or fatigue, body aches and maybe even a fever. . Sounds like some of those tries, maybe even having to stay home after work. “
“You hear some people in the trials who are disappointed that they don’t have any of these things, feeling that they must have had a placebo,” she added.
The committee meeting comes three days after Pfizer and its partner BioNTech applied for emergency use authorization from the Food and Drug Administration for their coronavirus vaccine.
The FDA process is expected to take a few weeks, and an advisory committee meeting to review the vaccine is scheduled for early December. Some Americans could receive their first dose of the vaccine in about a month.
ACIP should call an emergency meeting to make specific recommendations on distribution once the FDA clears a vaccine.
Federal agencies are already sending vaccination plans to staff. Five agencies have started telling employees they could receive the Pfizer or Moderna’s Covid-19 vaccine in as little as eight weeks, a person with first-hand knowledge of the projects told CNBC on Friday.
[ad_2]
Source link